MAP SPREAD TO WILDLIFE
2024-02-14 17:55:21 Researchers from Universidade de Lisboa, Lisboa, Portugal reported on the association of MAP infections in cattle and wildlife as well as MAP presence in soil in a small region of Portugal called Charneca do Infantado, Companhia da Lezíria. Their findings were published in the journal Environmental Pollution February 2024 (it is not Open Access).
Researchers from Universidade de Lisboa, Lisboa, Portugal reported on the association of MAP infections in cattle and wildlife as well as MAP presence in soil in a small region of Portugal called Charneca do Infantado, Companhia da Lezíria. Their findings were published in the journal Environmental Pollution February 2024 (it is not Open Access).
ABSTRACT
Mycobacterium avium subsp. paratuberculosis (MAP) is the etiological agent of paratuberculosis, a chronic infection affecting ruminants and other species worldwide. Information on the ecological factors that increase infection risk at the livestock-wildlife-environment interface remains scarce. Thus, this work aimed at determining which factors modulate the exposure of a mammal community within a Mediterranean agro-forestry farmstead to MAP. Through field, molecular and ecological modeling approaches, MAP prevalence, distribution and spatial risk at the livestock-wildlife-environment was estimated in the study area by screening 436 samples (cattle, n = 150; wildlife, n = 206; soil, n = 80). Using molecular detection of IS900 as proxy, MAP was identified in ten wild mammal species. Being a central prey of mesocarnivores in Portugal, the high prevalence of MAP in the wild rabbit (19%) may be related with red fox's (22%). MAP was also detected in cattle managed in the farmstead (animal and herd prevalence, 54% and 100%) and in soil (44%), which may perpetuate intraspecies and interspecies transmission. Wildlife diversity showed a positive influence on MAP presence in wild mammals, while wildlife abundance showed a negative effect. Land use variables exerted distinct degrees of impact upon MAP detection in specific groups of mammals: mixed forest cover showed positive influence on carnivores, and shrubland showed positive effect on wild rabbits. The prevalence of MAP in cattle showed a negative influence on the detection of MAP in lagomorph, which may stem from wild rabbit lower density and avoidance of cattle areas. Based on explanatory variables, the spatial prediction of MAP occurrence in wildlife indicated two hotspots with increased exposure risk but future studies are needed to confirm this projection.
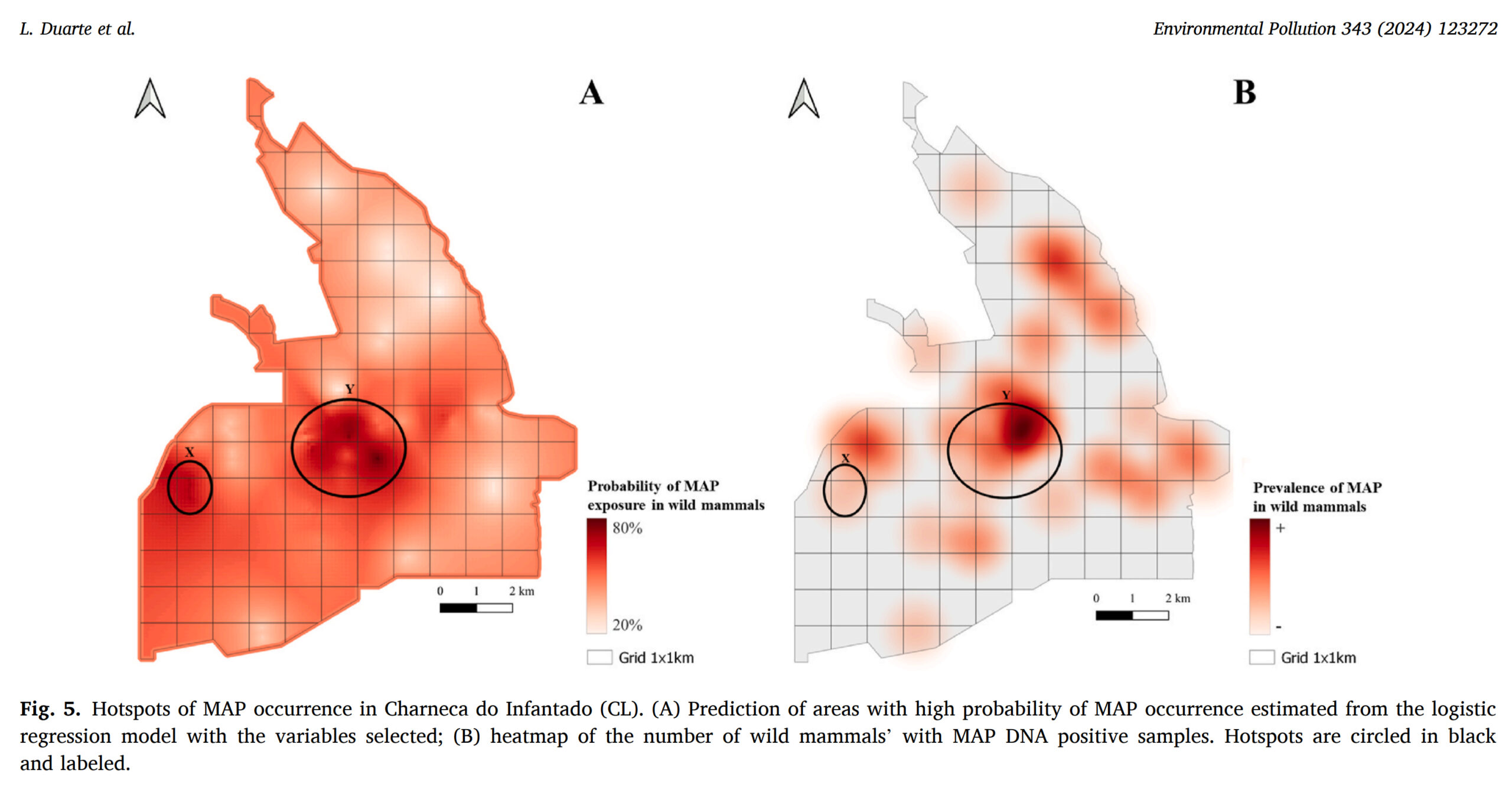
This work represents the most comprehensive molecular survey of MAP occurrence and determinants in Mediterranean agroecosystems leveraging the principles and tools of community ecology, debating potential biological and ecological effects underlying MAP transmission.
COMMENTS
This novel study reveals an association between the rate of MAP infection in cattle and wildlife. It remains to be determined if the infection spreads only one-way, i.e., cattle to wildlife, or both directions. Other studies have suggested that some wildlife are dead-end hosts (not passing sufficient numbers of MAP in feces to cause infection in other animals) while others, especially rabbits, can be sources of infection for livestock. The study detected MAP in the soil by IS900 PCR. As such, it is not possible to say of the MAP was alive or dead, and thus it’s impossible to know if MAP-contaminated soil is a risk for animal or human infection. Regardless, this study clearly illustrates that MAP is a concern for many more animals than livestock which heightens the importance of programs to control MAP.
Our website has more information specifically about MAP infections of wild ruminants, and both domestic and non-domestic nonruminants (including humans).
INTERNATIONAL COLLOQUIUM ON PARATUBERCULOSIS
2024-02-01 16:55:32
The upcoming 16th International Colloquium on Paratuberculosis will be held in the cities of Mathura and Vrindavan, India, October 21-25, 2024. Abstract submission is currently open and will close on March 31, 2024. Early bird registration ends on May 31, 2024. For more details about the conference venues, accommodations, and abstract submission please check the conference website.
MAP: A ZOONOSIS
2024-01-22 19:25:28Today’s news highlights the human health implications of MAP citing three different recently published articles. Shahzad and coauthors from Pakistan published a review article titled “Paratuberculosis; A Potential Zoonosis” in the journal Zoonosis (Open Access). Antonio and colleagues in Mexico describe detecting MAP in goat milk and various intermediate products in the production of cheese from goat milk (Journal of Veterinary Medicine and Animal Health, 2023 – Open Access) which is an example of how humans can be exposed to MAP even if they are not farmers. The third article is a book chapter written by C.T. Dow titled “Mycobacterium avium ss. paratuberculosis and Human Disease: Bridging Infection and Autoimmunity”. This chapter focuses on the fact that an immune response to a microbial pathogen like MAP can trigger a variety of human diseases that are labeled as autoimmune diseases.

ABSTRACT
Paratuberculosis, commonly known as Johne's disease (Yo’-nees), is primarily a disease of ruminants such as cattle, sheep and goats. It is a chronic infectious disease. The name of the disease is derived from the scientist’s name who discovered it in 1985 named Johne’s along with his colleague Frothingham. The disease is associated with Mycobacterium avium subspecies paratuberculosis (abbreviated as MAP) is an obligate intracellular organism. This bacterium mainly damages the intestines. MAP is a member of Mycobacteriaceae family which also includes M. tuberculosis and M. leprae, causative agents of tuberculosis and leprosy, respectively. Paratuberculosis is also known as “Silent slayer” in USA. The prevalence of this disease is continuously increasing every year due to lack of proper disease control programmes. The reasons behind could be lack of awareness in public as well as the lack of concern shown by respective governmental disease control authorities. Paratuberculosis is chronic in nature due to which there is no accurate treatment for it. The transmission source for this disease is the infected animal. There is an ongoing uncertainty regarding its transmission to human as various researches have produced contradicting results. Its prevalence rate varies in different regions of the world however; it is found most commonly in the countries having intense livestock farming. Crohn’s disease (CD) is the term used for the disease in human where the clinical symptoms are similar to those seen in Johne’s disease in animals. MAP is considered the primary cause of CD along with other associated factors however it is yet not confirmed. Primarily, the consumption of dairy products, obtained from infected animals, is considered its mode of its transmission to human. Actual burden of CD is yet unknown as it goes unreported in most of the countries due to lack of awareness among the people & lack of sufficient funding for research purpose. Interdisciplinary research collaborations are necessary to cover the knowledge gaps regarding paratuberculosis, highlighting the significance of surveillance and preventive measures to reduce possible health hazards to people.

ABSTRACT
Mycobacterium avium subspecies paratuberculosis (MAP) is the etiological agent of paratuberculosis, which is one of the chronic diseases that cause financial losses in livestock production. MAP can be present in cheese and other dairy products, especially those made with unpasteurized milk. Contamination of the food supply chain exposes humans to the bacteria, making the disease an important zoonosis of public health significance and a one-health emergency. The purpose of the study was to determine the presence of MAP in raw goat milk, natural rennet, and artisanal fresh cheese. A total of 18 milk samples were collected directly from the bulking tank, 23 from fresh cheese, and 10 from milk rennet from five municipalities in the State of San Luis Potosi, Mexico. Samples were analyzed through bacteriological culture and IS900 PCR-n. Statistical analysis was carried out in STATA® 7.0, analyzing frequencies and the Kappa test to determine the concordance index between bacteriological culture and IS900 PCR-n results. MAP was isolated from four milk samples (n=4/18, 22%), one from cheese (n=1/23, 4.3%), while none were obtained from rennet samples. IS900 PCR-n detected 22 positive samples: 6/18 (33.3%) in milk, 8/10 (80%) in rennet and 8/23 (34.74%) in cheese. Concordance between IS900 PCR-n and bacteriological culture in milk samples was high (0.7273) but low in cheese samples (0.0707). MAP was detected in milk and artisanal cheese, although it is noteworthy that MAP genomic material was detected in 80% of rennet samples analyzed with PCR. Quality control of milk, rennet, and all the inputs used for making cheese is necessary.

Abstract
Common to many inflammatory/autoimmune diseases is the concept of an “environmental trigger.” Mycobacterium avium subspecies paratuberculosis (MAP) is the known infectious cause of Johne’s disease, an enteric inflammatory disease mostly studied in ruminant animals. For years, MAP has also been implicated in the very similar Crohn’s disease of humans and sarcoidosis, a systemic granulomatous disease. More recently, MAP has been associated with juvenile sarcoidosis (Blau syndrome), autoimmune diabetes, autoimmune thyroiditis, multiple sclerosis, rheumatoid arthritis, and Parkinson’s disease. While it may be intuitive to implicate MAP in granulomatous diseases where the microbe participates in the granuloma, it is more difficult to assign a role for MAP in diseases where autoantibodies are a primary feature. MAP may trigger autoimmune antibodies via its heat-shock proteins. Mycobacterial heat-shock protein 65 (HSP65) is an immunodominant protein that shares sequential and conformational elements with several human host proteins. This chapter proposes that MAP is a source of mycobacterial protein antigens and acts as a trigger to an array of inflammatory and autoimmune diseases.
COMMENTS
All major mycobacterial pathogens are zoonotic, and yet medical science seems resistant to assigning this designation to MAP. Moreover, diseases can be triggered both by the direct effects of infection or by a misguided immune response leading to immune-mediated (autoimmune) diseases. Well known human examples for this autoimmune mechanism are Strep throat, caused by Group A Streptococcus sp., leading to rheumatic fever (heart damage) and food poisoning due to Campylobacter jejuni leading to Guillain-Barré Syndrome (nerve paralysis).
Lacking the zoonotic pathogen designation, it is hard for researchers to secure funding to study MAP as a human health concern. It is a circular problem: when MAP is not considered zoonotic by human health agencies it results in no research funding which in turn results in no clarity about this problem of potentially enormous human impact, and so we continue with the status quo. Likewise, until MAP is officially declared a zoonotic pathogen the financial burden to control MAP in food-producing animals, and thereby limit public exposure to MAP, rests largely on the shoulders of animal owners. Control of zoonotic pathogens should be a societal responsibility.
For more on the link of MAP and Crohn’s disease, visit this page of our website which contains at the end a long list of refereed scientific publications supporting the link, or watch the short presentation titled MAP is a Zoonotic Pathogen on this page.
JD CONTROL IN LARGE DAIRY CATTLE HERDS
2024-01-07 17:04:20Hungarian researchers reported on efforts to control paratuberculosis in 42 large dairy herds (average herd size 739 cows). This very practical study was reported in the January 2024 issue of Animals. The article is Open Access (free to everyone).
ABSTRACT
Paratuberculosis (PTB) is a severe, slow-developing, untreatable disease of ruminants. Worldwide, the disease affects more than 50% of herds in the dairy industry and causes substantial economic losses for dairy producers. Diagnostic tests show limited sensitivity, especially in the early stages of the disease. Our study aimed to investigate the seroprevalence of Mycobacterium avium ssp. paratuberculosis (MAP) in large-scale dairy herds in Hungary, in association with the self-reported presence or absence of screening and intervention measures against MAP transmission. We processed data from 42 large-scale Holstein Friesian farms in Hungary between 1 January 2018 and 31 December 2021. An average of 32,009 (min.: 31,702; max.: 32,207) animals were blood sampled yearly (127,372 in total during the four years), corresponding to 15% of the Hungarian dairy cattle population. All female cattle older than 2 years were blood sampled on the farms enrolled in the study. The samples were tested using a commercial ELISA (IDEXX paratuberculosis screening Ab test). Farm managers were interviewed about their on-farm diagnostic and intervention approaches using a uniform questionnaire, including questions on the level of awareness, frequency of ELISA and PCR testing, and their strategies for culling adult animals and reducing transmission to newborn calves. By comparing the annual rate of change in seroprevalence and the amount of change observed during the four-year period, we concluded that test-and-cull strategies implemented in parallel with newborn calf management that aimed at preventing MAP transmission were superior to test-and-cull strategies alone; moreover, fortifying culling decision making via additional ELISA and PCR tests is superior to using a single ELISA result. For farms that carried out a complex program with both “test-and-cull” and proper newborn calf management, there was a proportional reduction in apparent seroprevalence at an average of 22.8% per year. Fifteen of the sampled farms had no measures in place to control paratuberculosis. On these farms, the seroprevalence increased by 12.1% per year on average.
COMMENTS
The Hungarian study was an observational one, meaning that all the messiness of human behavior (herd managers, herd owners, and veterinarians, etc.) was included. Yet, the author’s careful study design and analysis made the findings credible, practical, and useful. This is one of the largest trials of its kind specifically focused on large commercial dairy cattle operations.
The study confirms that if owners are unaware of paratuberculosis in their herds, the infection rate continues to rise. Whole-herd testing and culling of test-positive cows alone, while helpful, is not sufficient to control paratuberculosis. Regular herd testing, with concrete actions taken with test-positive cows, must be paired with changes to herd management to limit the chances of MAP transmission for a control program to be truly successful. These findings are in full agreement with the computer simulation of paratuberculosis control in dairy herds published by Collins & Morgan in 1992 (Preventive Veterinary Medicine, 14:21-32) as shown in the graphic below (note the long time scale).
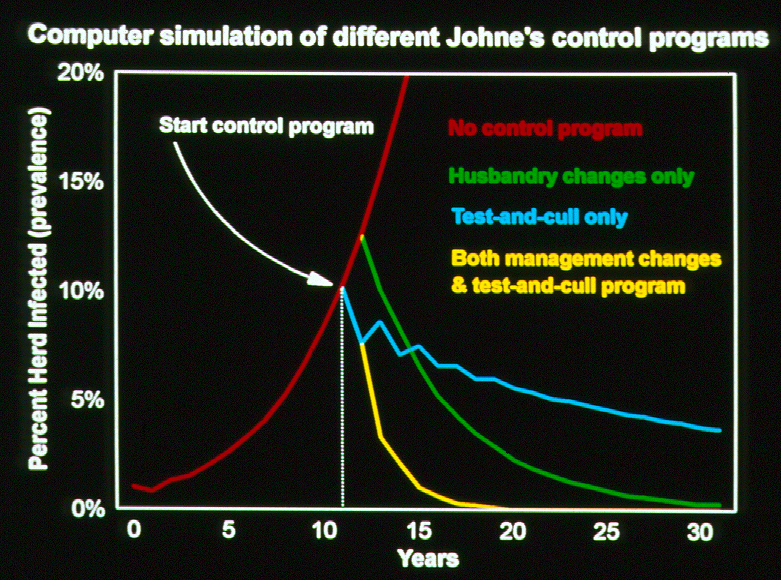
Over the course of this study, testing was voluntary and testing costs were subsidized by the state. As seen in many countries, when testing costs to herd owners are not subsidized, owners are far less likely to use diagnostic tests to control paratuberculosis. The impacts of this extend far beyond herd-level infection rates and herd productivity. Failure to control paratuberculosis in dairy herds means that a steadily increasing number of cows with become MAP-infected, more MAP will be found in raw milk and meat, and this will result in more MAP in the food supply with potential, if not probable, consequences for human health. The same holds true for other food-animal species affected by paratuberculosis. For more on this topic visit this page: https://johnes.org/presentations-and-mini-lectures/ and view the short presentation titled: MAP is a Zoonotic Pathogen.
JD RISK ASSESSMENTS
2023-10-19 16:20:59Researchers in Northern Ireland have analyzed the responses to a Veterinary Risk Assessment and Management Plan (VRAMP) concerning Johne’s disease for 1,569 dairy herds. Their findings are reported in the recent issue of Veterinary Record and the article is Open Access.

ABSTRACT
(British spellings)
Background
Animal Health and Welfare Northern Ireland has been enrolling dairy herds across Northern Ireland (NI) in a voluntary Johne's disease (JD) control programme since October 2020. A Veterinary Risk Assessment and Management Plan (VRAMP) questionnaire was completed for each herd enrolled and recommendations for improved farm management practices were provided to farmers. Herd JD testing was recommended but was not mandatory.
Methods
This study analysed VRAMP responses for 1569 dairy herds that had enrolled in the JD control programme up to October 2022. Univariate and multivariate regression models were applied to the data as appropriate.
Results
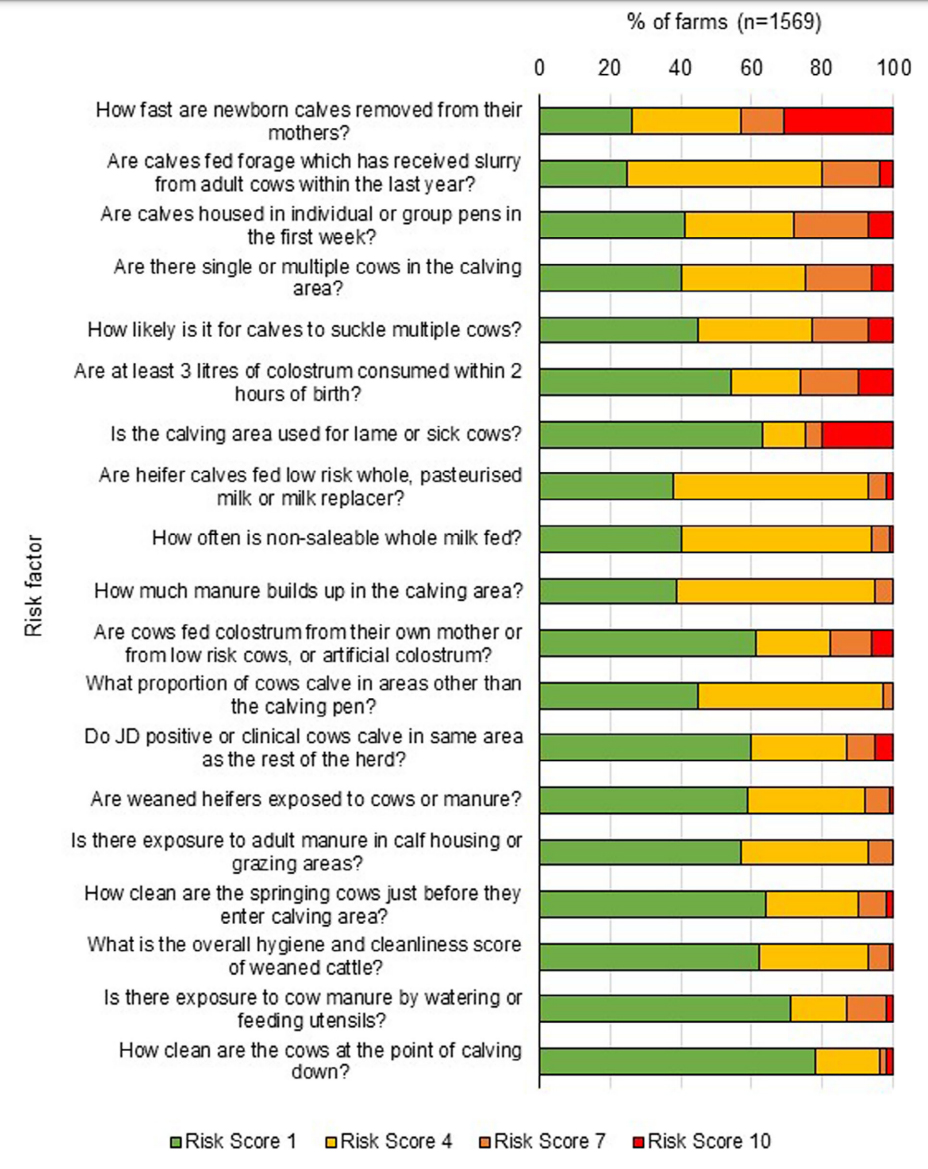
Overall, 21.4% of the dairy herds had completed herd JD screening, with 13.7% of herds reporting a confirmed case of JD. A further 31.5% of herds reported suspected case(s) of JD. Eighty-nine percent of farms had introduced animals from outside the herd. Herds that utilise a mixed calving pen and hospital pen, and herds that do not separate JD-positive or sick animals within the calving pen, were significantly (p > 0.001) more likely to be a high-probability JD herd. Accidental mixing of neighbouring herds significantly (p = 0.01) increased the risk of a suspected or confirmed case of JD. Herds that utilise rented land (70%) were significantly (p > 0.001) more likely to be at a high risk for JD.
Conclusions
The VRAMP analysis identified areas of JD control that should be focused on in NI dairy herds, such as calving pen management and hygiene. The results highlight the importance of common JD recommendations in the management of on-farm disease risk.
COMMENT
This report yet again illustrates that purchase of animals from other herds is a common and risky practice for the introduction of infectious diseases like Johne’s disease. The second common and risky practice on dairy farms is housing sick cows and JD test-positive cows in pens where calves are being born which further facilitates infection spread to the most susceptible animals on the farm and animals likely to join the herd as replacements in the future. These risky practices have been associated with higher MAP-infection rates in dairy herds in multiple studies in other countries.
Many countries have standardized risk assessment tools for Johne’s disease in dairy cattle herds and for beef cattle herds in some countries. In the U.S. the risk assessments for both dairy and beef herd can be done using an App for iPads making completion of the task quicker and providing the option to share the result with regulatory veterinary agencies. While similar risk assessments may not be available for goats and sheep, the same basic principles apply, and veterinarians can use these concepts to help animal owners develop the most effective JD control programs for their specific situation. Contact your local veterinarian.
MAP TRANSMISSION IN DAIRY HERDS
2023-10-06 16:25:18Researchers reported on an 11-year study of MAP transmission in 6 commercial dairy herds in the UK. Their Open Access publication appears in the October 2023 issue of Preventive Veterinary Medicine. The study is novel for its length and detailed analysis of MAP transmission. The findings largely support what has previously been reported and helps focus producers and veterinarians on the best control measures.

ABSTRACT
Johne's disease (JD) is a chronic disease of ruminants endemic in the UK and other countries and responsible for large economic losses for the dairy sector. JD is caused by Mycobacterium avium subspecies paratuberculosis (MAP), which typically infects calves that remain latently infected during a long period, making early detection of infection challenging. Cow to calf transmission can occur in-utero, via milk/colostrum or faecal-orally. Understanding of the different transmission routes to calves is important in informing control recommendations. Our aim in this longitudinal study was to measure the association between the transmission routes via the dam and the environment on a calf subsequently testing serologically positive for MAP. The study population comprised of 439 UK dairy calves from 6 herds enrolled between 2012 and 2013. These calves were followed up from birth until 2023. At birth individual calf data was captured. During follow-up, individuals entering the milking herd were quarterly tested for the presence of MAP antibodies using milk ELISA. Cox regression models were used to measure the association between exposure from the dam (in-utero and/or colostrum) or from the environment (long time in dirty yard) and time to first detection of MAP infection. An association between calves born to positive dams and probability of having a MAP positive test result remained after excluding potential MAP transmission via colostrum (Hazard ratio: 2.24; 95% CI: 1.14 – 4.41). Calves unlikely to be infected with MAP via the in-utero or colostrum route, had 3.68 (95% CI: 3.68 1.45–9.33) higher hazard of a positive test result when they stayed longer in a dirty calving area. The effect of the dam infection status on transmission to calves precedes the dam's seroconversion and persists after excluding the potential role of transmission via colostrum . The association between time spent in a dirty calving area and probability of a MAP positive test result highlights the role of environmental contamination as a source of infection in addition to the dam.
COMMENTS
This study is a noteworthy contribution to understanding the epidemiology of MAP infections in commercial dairy herds. The findings are consistent with those from other countries and the article provides a concise list of pertinent references. While this scientific publication is rather technical in nature, due to the nature of the statistical analysis, the “take home messages” are clear:
- MAP infection risk is partly driven by the dam’s infection status.
- Calves from infected dams have higher MAP infection risk, regardless of dam's test status at calving.
- Spending prolonged time in a dirty yard increases the risk of MAP infection.
- Dam's impact on MAP risk extends beyond colostrum transmission.
- MAP persistence in commercial dairy herds results from a combination of dam-related and environment-related factors.
JD IS COMMON IN GOATS.
2023-07-25 14:59:23July 18, 2023, Italian researchers published a study on paratuberculosis in 33 dairy goat farms in northern Italy. Their study explored associations between animal welfare and herd biosecurity with the rate of MAP infections in each herd as detected by a commercial serum ELISA kit. The study was published in the journal Animals and is Open Access.

ABSTRACT
Paratuberculosis is a notable infectious disease of ruminants. Goats appear to be particularly susceptible. The survey aimed to investigate the spread of paratuberculosis in Italian goat farming and evaluate whether the presence of the disease could be influenced by welfare and biosecurity deficiencies. A serological survey for paratuberculosis in 33 dairy farms in northern Italy was conducted. Contextually, animal welfare and biosecurity were assessed, using a standardized protocol of 36 welfare indicators and 15 biosecurity indicators which assigns to each farm a welfare and biosecurity score from 0 (any application) to 100% (full application). An overall result of less than 60% was considered insufficient. Nineteen farms (58%) tested positive for paratuberculosis, with a mean intra-herd seroprevalence of 7.4%. Total welfare ranged from 39.56 to 90.7% (mean 68.64%). Biosecurity scores ranged from 10.04 to 90.01% (mean 57.57%). Eight farms (24%) showed poor welfare conditions (welfare score < 60%) and 19 (58%) an unsatisfactory biosecurity condition (biosecurity score < 60%). With respect to the explorative character of the study, an indicative association between seven welfare and biosecurity indicators and paratuberculosis seropositivity was identified. The presence of paratuberculosis in northern Italy dairy goat farms was confirmed. The welfare and biosecurity assessment protocol proved to be an accurate tool, capable of identifying critical points for managing health, welfare and productivity.
COMMENTS
Paratuberculosis is common among goat herds and yet it is an often-overlooked infectious disease problem that compromises animal health, animal productivity, and animal welfare. The finding in Italy that 58% of the 33 herds tested were positive by ELISA is consistent with similar studies in other parts of the world. Importantly, they showed a correlation between the MAP-infection status of herds and their levels of animal welfare and herd biosecurity.
The Italian findings are consistent with other such surveys of goat herds in other countries done using commercial ELISA kits. In France, researchers tested 11,847 goats over 6 months old in 105 herds finding that 55.2% of herds were seropositive with 5.9% of goats being ELISA-positive within the MAP-infected herds (Mercier, 2010). In the U.S., Pithua reported in 2012 that 9 of 25 (36%) Boer goat herds in the state of Missouri were positive for paratuberculosis using a commercial ELISA kit. A 2011 study done in Cyprus tested 72 herds that had goats only or goats kept together and sheep with 4,582 total goats tested using a commercial ELISA kit. They found that 36 of 72 (50%) of herds with goats were ELISA-positive (1 or more positive goats). Within these ELISA-positive herds, 10.3% of goats tested ELISA-positive. A study of 116 herds or Korean Black Goats found regional differences in S. Korea for the rate of ELISA-positive goat herds ranging from 18.2% to 38.2% (Lee et al., 2006). A study of goat herds in the province of Ontario Canada published in 2016 found that 16 of 29 (55.2%) randomly selected dairy goat herds were ELISA-positive.
These high goat herd infection rates are all based on commercial ELISA kits and so can legitimately be compared. Because the ELISA is less sensitive than other diagnostic tests like fecal PCR, the reported values, called apparent prevalence rates, are underestimates. The true infection rates (called true herd-level prevalence) are even higher. In the Canadian study, the ELISA detected only 15/29 (52%) of all goats that were shedding MAP in their feces.
Clearly, paratuberculosis is a problem that needs to be addressed among goat herds globally for the sake of the animals, the various types of goat industries, and consumers of goat meat and dairy products.
To limit the risk of bringing MAP infections into goat herds, owners are advised to have a good biosecurity plan.
Specifically:
- Assume any goat herd you buy from is MAP-infected until proven otherwise. You will be right over 50% of the time. Some owners are simply ignorant about Johne’s disease. Others willfully ignore the problem by intentionally never testing.
- Only buy or lease goats from herds that are 100% test-negative for paratuberculosis (all adult goats in the herd) within the past 12 months. ELISA testing is acceptable but testing by fecal PCR is far more accurate, as the Canadian study showed.
- Quarantine all purchased animals until a repeat test is done by fecal PCR and found to be negative.
PREVENTION PAYS!
MAP IN CROHN'S DISEASE (AGAIN)
2023-06-11 15:52:58A team of 11 Portuguese scientists and one MAP expert from the UK have yet again demonstrated live MAP in the blood of Crohn’s Disease (CD) patients and confirmed the association of MAP and CD. In addition, they showed that MAP was far more common in CD patients than was Escherichia coli bacteria with adherence/invasive ability (AIEC). This type of E. coli has been suggested by some scientists to be a causal agent for CD. This work was published June 7 in the journal Microoganisms.
ABSTRACT
Pathobionts, particularly Mycobacterium avium subsp. paratuberculosis (MAP) and Escherichia coli isolates with adherence/invasive ability (AIEC) have been associated with inflammatory bowel disease (IBD), particularly Crohn’s disease (CD). This study aimed to evaluate the frequency of viable MAP and AIEC in a cohort of IBD patients. As such, MAP and E. coli cultures were established from faecal and blood samples (with a total n = 62 for each) of patients with CD (n = 18), ulcerative colitis (UC, n = 15), or liver cirrhosis (n = 7), as well as from healthy controls (HC, n = 22). Presumptive positive cultures were tested by polymerase chain reaction (PCR), for a positive confirmation of MAP or E. coli identity. E. coli-confirmed isolates were then tested for AIEC identity using adherence and invasion assays in the epithelial cell line of Caco-2 and survival and replication assays in the macrophage cell line of J774. MAP sub-culture and genome sequencing were also performed. MAP was more frequently cultured from the blood and faecal samples of patients with CD and cirrhosis. E. coli presumptive colonies were isolated from the faecal samples of most individuals, in contrast to what was registered for the blood samples. Additionally, from the confirmed E. coli isolates, only three had an AIEC-like phenotype (i.e., one CD patient and two UC patients). This study confirmed the association between MAP and CD; however, it did not find a strong association between the presence of AIEC and CD. It may be hypothesized that the presence of viable MAP in the bloodstream of CD patients contributes to disease reactivation.
COMMENTS
This study adds to the body of literature on the association of MAP and CD and strengthens the argument that MAP is a zoonotic pathogen, i.e., one transmissible from animals to humans. For more about MAP as a zoonotic pathogen and its link to Crohn’s Disease, see this page on our website where a long list of references on the subject is provided, with direct links to the original publications.
JD PATHOLOGY IN GOATS
2023-05-27 15:20:53E. Stefanova and nine colleagues in Spain have published an article on Johne’s disease pathology in goats. They contrast the pathology found in non-vaccinated goats and goats that have been vaccinated with a commercial heat-inactivated vaccine for Johne’s disease. They also describe lesions caused by bronchopneumonia and caseous lymphadenitis (CLA) plus pathology caused by assorted other disease problems. The pictures of gross pathology are particularly good. Mesenteric lymph nodes were a the most valuable tissue to sample for detection of MAP infections in goats by histopathology. This Open Access article was published in the journal Animals on May 19, 2023.
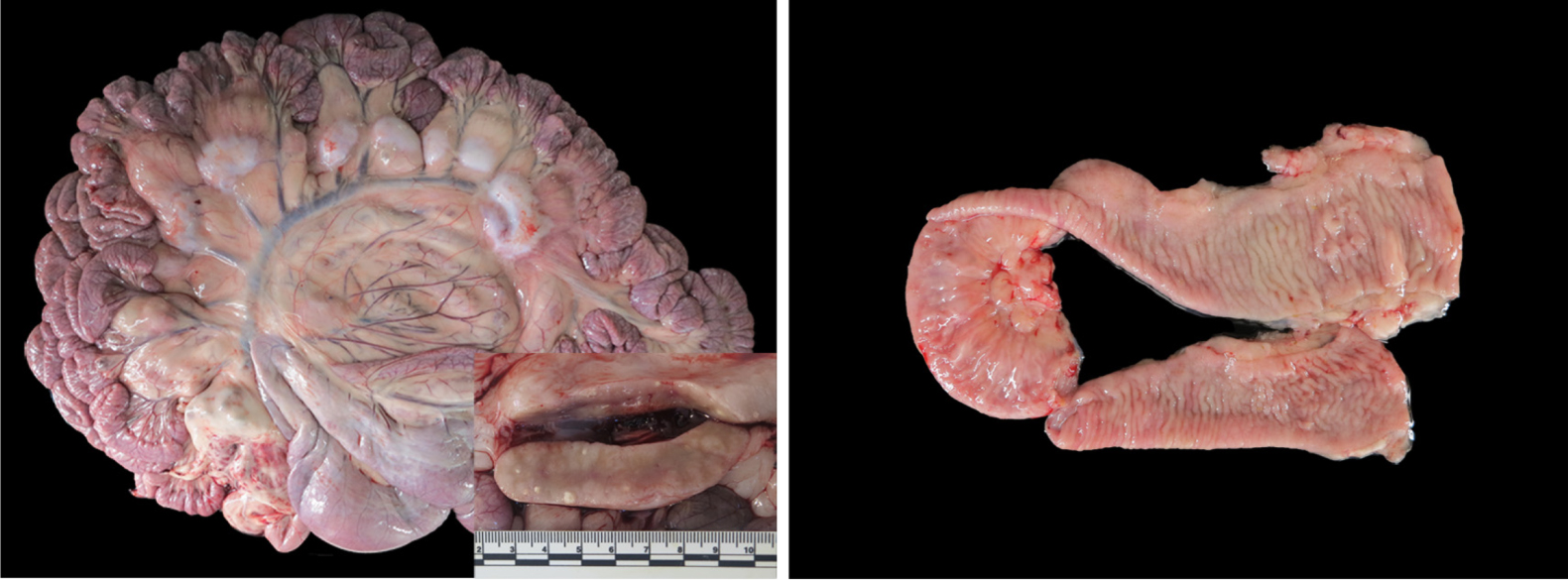
SUMMARY
Paratuberculosis (PTB), also called Johne’s disease, is a well-known disease with considerable financial impact on the farm industry worldwide. Nevertheless, data regarding the assessment of naturally infected goat herds is limited. The present study describes in detail the observed gross and histological lesions detected in 39 necropsies of goats (15 vaccinated and 24 non-vaccinated) from herds with a confirmed history of PTB. PTB microscopic lesions of various grades were detected in all animals in target organs and the presence of the causative agent was confirmed using different laboratory tools. The main inflammatory findings affected the hemolymphatic, respiratory and gastrointestinal systems. The lesions were confirmed microscopically with lesser macroscopically visible alterations. Our result demonstrated that non-vaccinated animals presented more severe PTB intestinal lesions and had respiratory inflammation in all age groups studied. Those also presented a higher prevalence of ileocecal valve PTB lesions. Gastrointestinal non-PTB lesions were detected in higher number in non-vaccinated goats. Thus, histology is a powerful tool for herd diagnosis and assessment. Mainly inflammatory lesions of the respiratory and gastrointestinal tract were detected in the studied PTB-affected herds. Additionally, vaccination against PTB could play a key role in the reduction of lung and gastrointestinal inflammatory processes present in the herd.
COMMENTS
The Gudair® vaccine used in this study is a commercial heat-inactivated vaccine containing 2.5 mg/mL of MAP strain 316 F with mineral oil adjuvant (CZ Vaccines S.A., Spain). In Spain, it is primarily used in sheep and goats. This vaccine is not licensed for use in the United States.
CONTROL PROGRAMS WORK!
2023-04-26 16:23:41 R. Scarpelli together with seven Italian colleagues reported on the success of the Voluntary National Control Plan and Guidelines developed by the Italian Ministry of Health in 2013 at controlling paratuberculosis in Italian dairy herds of varying sizes.
R. Scarpelli together with seven Italian colleagues reported on the success of the Voluntary National Control Plan and Guidelines developed by the Italian Ministry of Health in 2013 at controlling paratuberculosis in Italian dairy herds of varying sizes.
Their publication appears in the June 2023 issue of Preventive Veterinary Medicine.
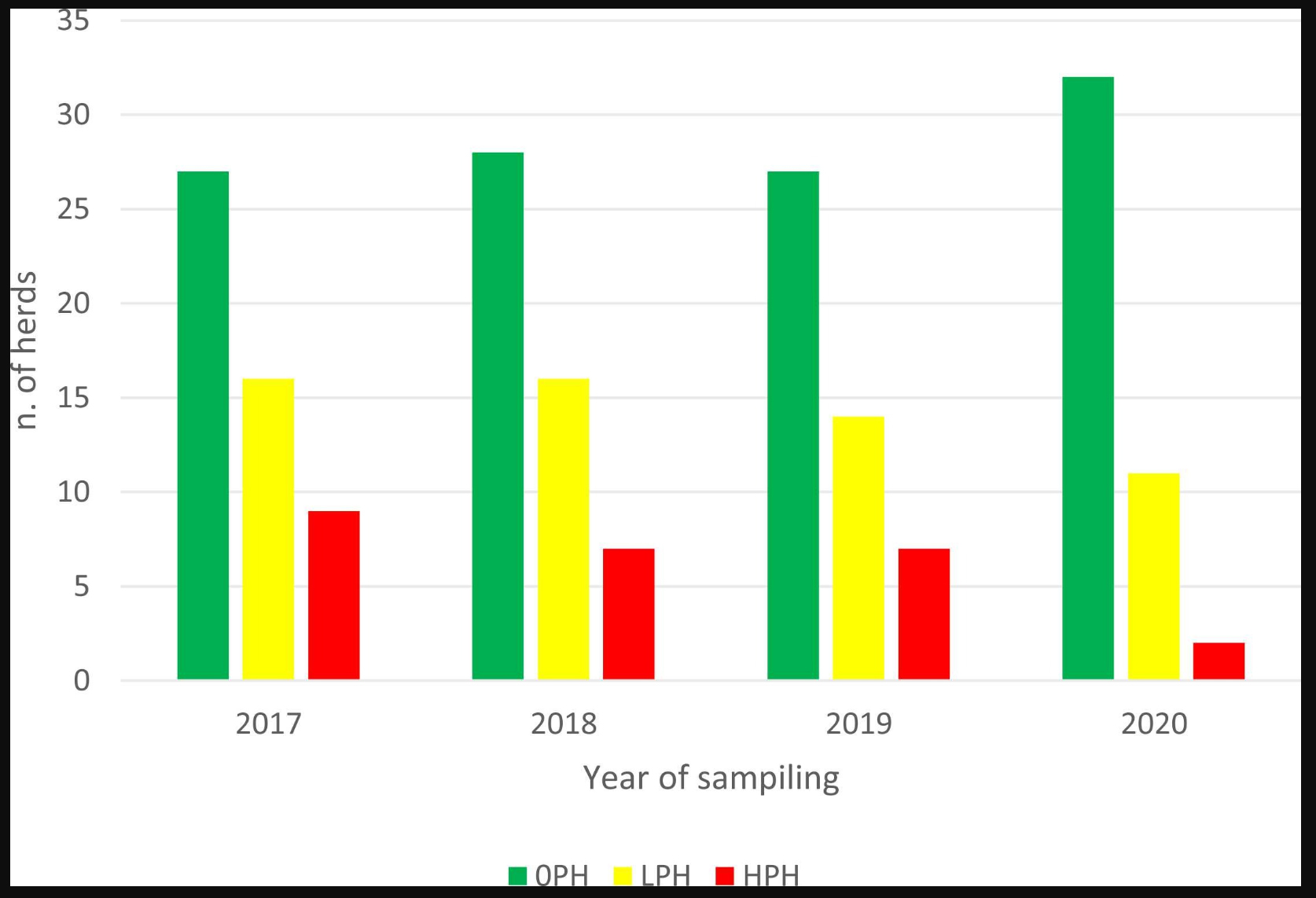
This graphic from their publication shows a steady rise in the number of ELISA negative herds (green bars), and a concomitant decrease in the number of herds with a low percentage of ELISA-positive animals (yellow bars) and number of herds with a higher percentage of ELISA-positive cows (red bars) over the time period of the study, 2017 to 2020. Interestingly, one farm that decided not to follow the proposed control program, but accepted to be sampled every year, showed an increasing rate of ELISA-positive cows (from 5.2% in 2017 to 7.6% in 2020).
ABSTRACT
Paratuberculosis is considered one of the most economically devastating infectious diseases of domestic livestock, and the most effective control strategy is a combination of ‘test-and-cull’ and on-farm biosecurity measures. In Italy, a Voluntary National Control Plan (VNCP) and guidelines have been introduced to reduce the impact of the disease, and farmers can voluntarily enroll in the control plan. The main aims of this study were: i) the description of the trend over a 4-year period on total, within-herd (WH) and between herd (BH) apparent seroprevalences observed in 64 dairy herds members of a mutual company located in Italy after the introduction of a proposed “Customized Control Plan” (CCP); ii) the evaluation of its effectiveness in terms of percentage of participating farms that decided to join the VNCP. Analyses on serum samples were performed with Enzyme-Linked ImmunoSorbent Assay (ELISA) method and revealed a general decrease in both total, WH and BH apparent seroprevalence. Total average apparent seroprevalence decreased from 2.39% in 2017 to 1% in 2020. Negative herds rose from 51.9% in 2017 to 71.1% in 2020, while farms with WH apparent seroprevalence >5% decreased from 17.3% in 2017 to 4.4% in 2020. BH apparent seroprevalence decreased from 51.2% in 2017 to 29.2% in 2020. Among the 52 out of 64 herds that accepted to continue the proposed CCP after the first year, 41 (78.8%) joined in 2020 the VNCP, that assessed the health ranking of the herds. The results provide evidence that a control plan based on a farm-specific strategy and a subsidized testing process can effectively reduce the impact of paratuberculosis in dairy herds, especially in convincing farmers to continue in paratuberculosis control by joining the VNCP, including them in a national context and increasing their awareness of the disease.
COMMENT
This study adds to the body of literature showing that paratuberculosis (Johne’s disease) in dairy cattle herds can be controlled using a cost-effective diagnostic test (ELISA) combined with herd management changes.
« Previous 1 2 3 4 … 18 Next »